A Look into the Past
“The infant’s age, Mrs. Lancaster?” The ophthalmologist deftly grasped the squirming baby, shining a light into his eyes.
“Twelve months old, Dr. Tay.” The child’s gaze slowly followed the light’s movement, and he let out a loud wail of discomfort.
“Have you suspected anything abnormal about him?”
“I’ve—I’ve seen that he isn’t as quick to notice things as other babies, and he’s almost too weak to hold his head up,” she whispered. “Nothing seemed wrong until he just got…so weak.”
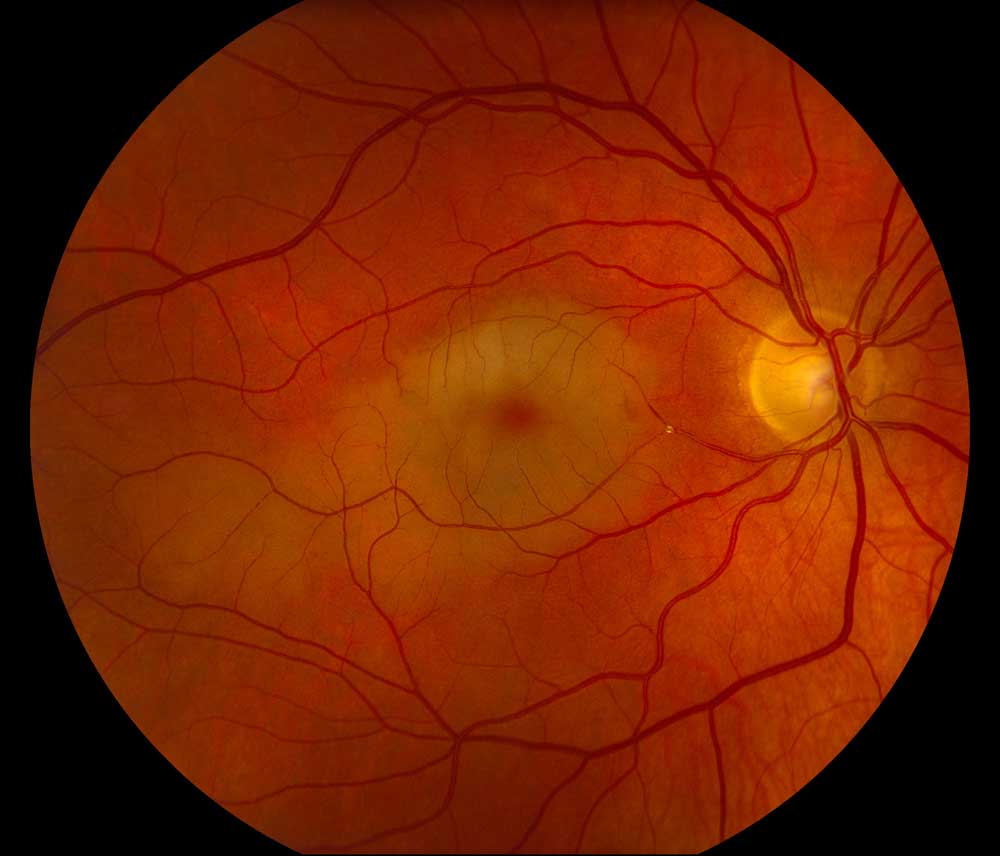
Peering at the child’s eyes again, Dr. Warren Tay observed a strange phenomenon that he would later describe in an 1881 medical journal as a “mahogany-colored, circular spot contrasting sharply with the surrounding white of the eye.”
While he struggled to understand the baby’s strange condition, Tay could only watch as the child grew increasingly weaker over the next few months. As other babies started speaking and walking, this child became quieter and quieter, emitting only the occasional gurgle. Eventually, he lost all control over his limbs and could not swallow or see. He died before his fourth birthday.
The “cherry-red spot” would be become one in a list of identifying factors for the rare disease first studied by Tay and later, the American neurologist Bernard Sachs. However, neither would fully understand the mechanisms behind the disease known today as GM2 gangliosidosis or infantile Tay-Sachs disease.
One Hundred Years Later
Tay-Sachs disease is a genetic disorder caused by the absence of the vital protein Hexosaminidase A isoenzyme (Hex-A). Hex-A breaks down fatty substances called GM2 gangliosides. However, infants with Tay-Sachs possess a gene mutation that makes them unable to produce Hex-A. As a result, fatty substances, or lipids, quickly accumulate inside neurons causing severe nerve damage (Sargeant et al., 2012).
The rapid deterioration of the nervous system poses a challenge to researchers. How can they cure a disease that they cannot monitor and study for long periods of time?
The answer could lie with an animal that has been around since biblical times, a rare breed of spotted black-and-white sheep—the Jacob sheep.
Sheep are well-known to insomniacs; they are the fluffy, gentle creatures we count when we cannot sleep. As it turns out, sheep – specifically, Jacob sheep – now have a much larger purpose in the field of research.
In 1999, two sheep breeders, Fred and Joan Horak, observed a peculiar condition among their flock of Jacob sheep (National Tay-Sachs & Allied Diseases, n.d.).
A couple of the lambs began to totter and lose their mobility, eventually dying. After consulting a perplexed veterinarian, the Horaks looked to researchers at Texas A & M University for answers (Geshelin, n.d.). Still, years went by without conclusive results.
Researchers from various universities joined efforts to autopsy four Jacob sheep that had died of symptoms similar to those of the Horak’s lambs. After discovering an absence of the Hex-A activity and an unusually high concentration of gangliosides in the brains, the scientists realized that the Jacob sheep had Tay-Sachs disease.
The discovery presented an unexpected opportunity to test a potential gene therapy for the treatment of Tay-Sachs in humans (Friedman, 2010). Previously, researchers had found the disease in flamingos and penguins. However, not only did flamingos and penguins prove difficult and expensive to care for, but they also produced offspring infrequently (Torres et al., 2010).
“They [Jacob sheep] mate every year, and they’re sexually mature at one year old,” said Dr. Edwin Kolodny, a member of the aforementioned research group, in 2010.
Most importantly, Jacob sheep have a remarkable 86% DNA similarity with humans, which is more than flamingos or penguins do. Because of this genetic similarity, the clinical and biochemical features of the Jacob sheep model are likely to mirror those of the human infantile form of Tay-Sachs well (Torres et al., 2010). This means that treatments like gene therapy that are carried out successfully in Jacob sheep are much more likely to succeed in humans.
Despite many breakthroughs, scientists still do not understand the process by which the accumulation of gangliosides affects neuronal function. Promising treatments such as gene therapy are in development, but none has been clinically approved yet (Sargeant et al., 2012).
The Jacob sheep is the first viable large animal model to be used in the study of Tay-Sachs disease. We are counting on these sheep to save families from the nightmare of watching their beloved babies die from a genetic disease for which there is still no treatment or cure.
In Brief:
- Tay-Sachs is a rare inherited disease caused by a genetic mutation.
- Jacob Sheep, like humans, can be born with Tay-Sachs.
- Jacob Sheep are easier to raise and study in the lab than other animals such as flamingos and penguins.
This article was written by cYw16. As always, before leaving a response to this article please view our Rules of Conduct. Thanks! -cYw Editorial Staff


![[Photo: "Jacob Sheep Looking at Camera", License: iStockphoto]](https://www.curiousyoungwriters.org/wp-content/uploads/2014/02/iStock_000005557774_Small.jpg)