As children, we were all prodded to “eat our fruits and vegetables.” But in the future, will we be injecting plant life as well? Based on several discoveries to date, this very well may happen. Plants can be used in medications, including vaccines.
Although plants also have numerous health benefits as a food source, many may be unaware that they are also frequently used in medications. They were even utilized as herbal treatments to suppress COVID-19 replication (i.e. the Japanese honeysuckle, Chinese Rhubarb and Siberian Apricot). However, they aren’t regularly used in vaccines. That reality may be changing over time.
There are a couple of very good reasons why pharmaceutical companies might wish to turn to plants. Plant therapeutics can have a low infrastructure cost. They can also at times, be produced more swiftly than the traditional development process.
Plants that might be used for vaccine development include rice, tobacco, lettuce and potatoes, which are all readily available. Vaccines produced using modern methods commonly employ acids, sugars or RNA as key ingredients, which have a record of being potent, but can be expensive to produce. Despite the many promises of plant-based vaccines, there have been few cases of them being developed. When looking at pharmaceutical companies, unrelated to big pharma, it seems plant vaccines are still in their early stages.
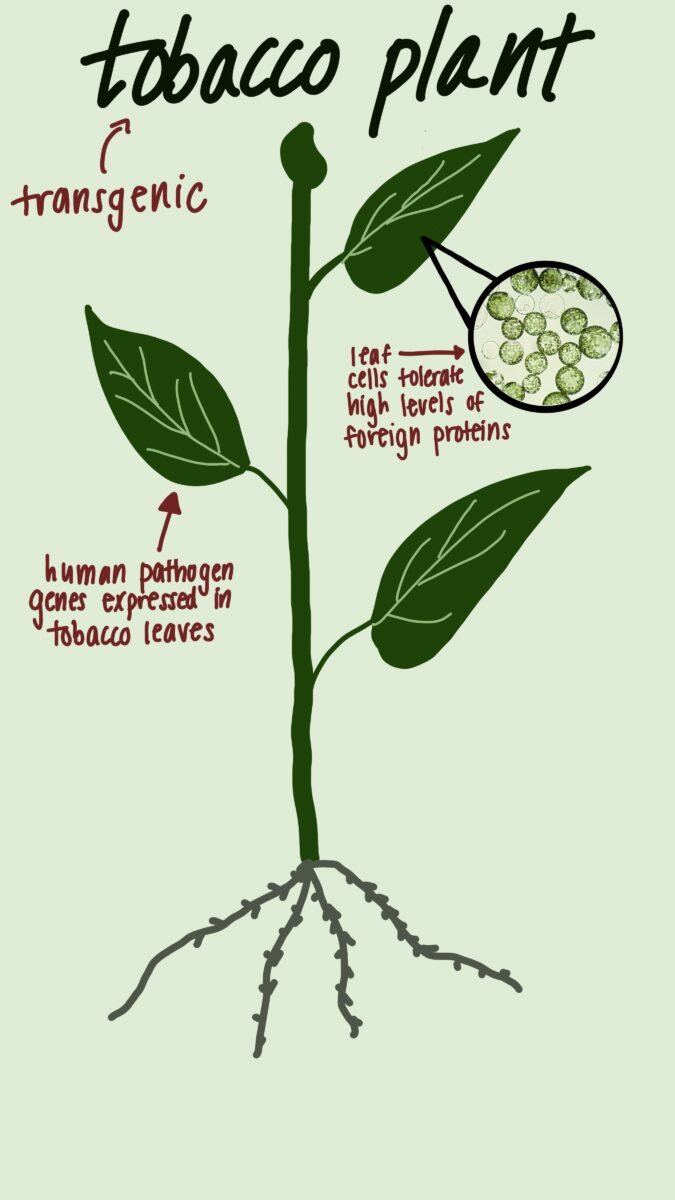
Medicago, a Quebec-based biotechnology firm is one of the few companies known for creating plant-based vaccines. Before the company closed its doors earlier this year, their products featured ingredients such as “Nicotiana benthamiana,” a plant related to tobacco, in the development of the company’s promising but now shelved COVID-19 vaccine: Covifenz™. This plant’s ability to tolerate high levels of foreign proteins in its leaf cells makes it one of the most widely used plants for molecular studies.
Brian Ward, M.D., a professor of medicine and microbiology at McGill University and a former medical officer at Medicago, was a trailblazer in the development of Covifenz. He says the vaccine was produced by unconventional means but contained the typical protein “therapy” visible in other vaccines. The plant’s recombinant – or genetic – character helps accelerate the vaccine development timeline, which is critical in an urgent pandemic situation.
Plant vaccines have been shown to be successful in combating infectious diseases in the past. Israeli researchers have already received FDA approval for “Protalix.” This vaccine for Gaucher disease, a genetic disorder that creates immense abdominal pain and anemia caused by the lack of glucocerebrosidase, contains the same tobacco-related plant as Covifenz and has been administered with positive results. Similarly, Merck received FDA approval for its Ebola vaccine which uses rice proteins and is now noteworthy for its worldwide use.
While Merck was developing its Ebola vaccine, Kentucky Bioprocessing, along with drugmaker Defyrus, started its Ebola vaccine research and development in 2014 using Nicotiana.
According to a marketing officer from Kentucky Bioprocessing, “… manufacturing ZMapp antibodies in tobacco variety Nicotiana Benthamiana …produces plant-based proteins in a way that is faster and more easily scalable than bioreactor-based methods,” which use mini reactors to carry out biological operations on a big scale.
While these examples show the promise of plant-based vaccines, much remains to be considered and improved. Due to longstanding concerns about genetically modified food crops, public perception of altered plants is one area for consideration and education.
The laws regulating plant use also differ from those of the widely accepted Good Manufacturing Practices, so it’s important for the public to understand the integrity of the production process. Part of this process involves selecting the right antigens, a scientific word for foreign substances that produce immune responses. Some antigens aren’t compatible with certain host plants, which makes the pre-trial production process longer. And just like other foods, plant therapeutics can trigger allergies, so that must also be assessed during development, safety testing, and production.
Dr. Ward acknowledges “there are some political and perception issues in using [tobacco and/or tobacco-related plants] to produce a vaccine,” and adds “most people are inclined to think recombinant protein expression in a plant is more natural than other common expression systems, like yeast, bacteria or mammalian cells.”
Nevertheless, he thinks the status quo is already changing and believes the coming years will bring even more plant-based treatments and vaccines to improve human health.
- Although plants also have numerous health benefits as a food source, many may be unaware that they are also frequently used in medications.
- Plants that might be used for vaccine development include rice, tobacco, lettuce and potatoes.
- However, due to longstanding concerns about genetically modified food crops, public perception of altered plants is one area for consideration and education.
Sources
Takeyama, Natsumi. “Plant-based vaccines for animals and humans: recent advances in technology and clinical trials.” National Library of Medicine. Last modified on September 3rd, 2015. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4667769/
“Plant-Derived Vaccines.” Biopharmadealmakers,” https://www.nature.com/articles/d43747-020-00537-y
Pharm, Ratan. “Plant-Derived Vaccines.” News Medical Life Sciences. Last modified on July 22nd, 2021. https://www.news-medical.net/health/Plant-Derived-Vaccines.asp
Hensley, Laura. “A Plant-Based COVID-19 Vaccine Could Soon Hit the Market.” VeryWell Health. Last modified on February 27th, 2022. A Plant-Based COVID-19 Vaccine Could Soon Hit the Market (verywellhealth.com)
“Tobacco Plants Contribute Key Ingredient For COVID-19 Vaccine.” NPR. Last modified on October 15th, 2020. https://www.npr.org/sections/health-shots/2020/10/15/923210562/tobacco-plants-contribute-key-ingredient-for-covid-19-vaccine
LeBlanc, Zacharie. “Plant-Based Vaccines: The Way Ahead?” National Library of Medicine. Last modified on December 22nd, 2020. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7822169/
Barry, Fionna. “CMO Kentucky Bioprocessing Grows Ebola mAb from Tobacco.” Outsourcing-Pharma.com. Last modified on September 2nd, 2014. https://www.outsourcing-pharma.com/Article/2014/09/02/CMO-Kentucky-Bioprocessing-grows-Ebola-mAb-from-tobacco
Editorial Team
- Chief Editor: Juhi Amin
- Team Editor: Audrey Yan
- Creative Team Senior Managers: Daniela Benoit, Bebe Lemanowicz
- Creative Team Manager: Annika Singh
- Social Media Team Managers: Spencer Lyudovyk, Yoojin Jeong
- Image Credits: Annika Singh
Mentor
Lisa Newbern is Chief of Public Affairs for the Emory National Primate Research Center at Emory University. In this role, she provides strategic counsel, develops measurable action plans and leads teams in executing communication programs that include executive messaging, issues and risk management, media relations, internal communication, community, educational and collaborative outreach, social media, online presence, government relations, special events, sponsorship opportunities, fundraising and historical archiving. She also leads the collaborative public relations initiatives for the seven NPRCs, including NPRC.org and @NPRCnews. Among her service activities, she is a board member for Americans for Medical Progress; a member of the Georgia Department of Education Division of Special Education State Advisory Panel and a mentor to students in Emory University’s Genetics Counseling Program.
Content Expert
Dr. Brian Ward, Ph.D., is a professor at McGill University in Quebec, Canada, instructing students in medicine and microbiology. He also previously served as a medical officer at Medicago: a Canadian based pharmaceutical company. His expertise lies in RNA viruses, vaccine research and development, and international health. At the start of the COVID-19 pandemic, he played a critical role in developing Medicago’s plant-based vaccine.